Hesperian Health Guides
Learn about chemicals used in your factory
HealthWiki > Workers' Guide to Health and Safety > Chapter 8: Chemical dangers > Learn about chemicals used in your factory

You have a right to know what chemicals you work with. By learning more about the chemicals, you can seek medical or professional help, you can organize for safe chemicals with your coworkers, and together you can work with your employer to reduce exposures and eliminate toxic chemicals from production.
Contents
Talk to other workers
Ask workers what they know or can find out about the chemicals in the workplace. Collect all the chemical names: brand names, generic names, and even nicknames. Write down any characteristics that can help identify the chemical, such as how and where it is used, its color, smell, and any instructions the employer gave about how to handle the chemical ("Always put the chemical into water, not the other way around!"). Talk with the workers in the shipping and receiving areas, and the people who inventory, store, mix, and dispose of the chemicals and their containers. They often know the names of the chemicals, or can find out.
Write down any health problems you or others feel while working with or transporting the chemicals. Ask: Do you feel ill at work or after work? Are some work areas better or worse than others? Do you feel better when you are away from work for a few days?

What chemical is it?
If you know the name of the chemical, you can usually find out about its health effects by looking up information about it. But if you don’t know its name, you may be able to find out what it is by its color, smell, what it is used for, and other qualities. For help in doing this and to understand the dangers of specific chemicals, see the Appendix B: Common chemicals and materials.
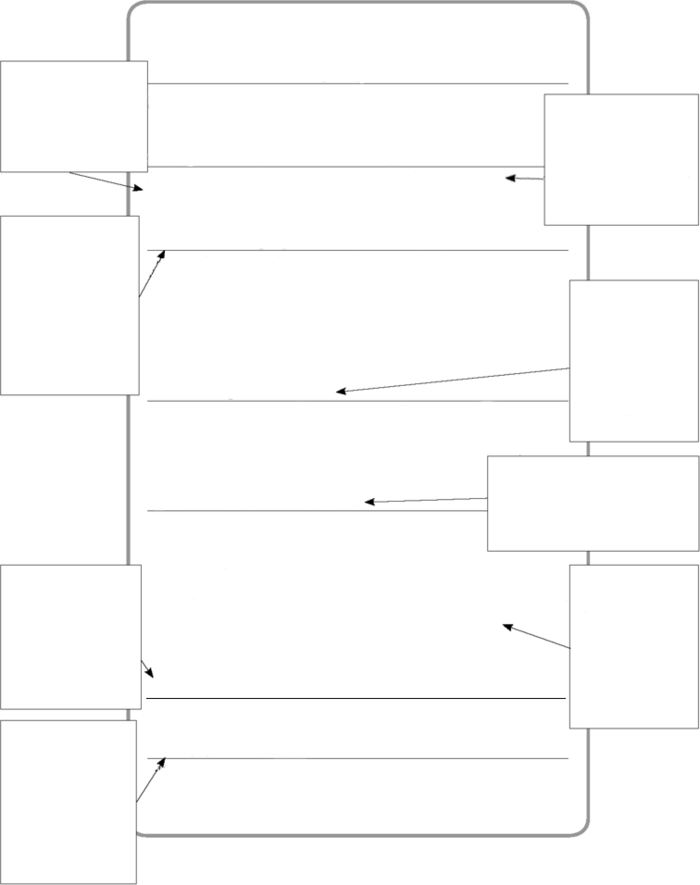
| ISOPROPANOL (IPA) Contains: Isopropyl alcohol . . . . 70% |
| DANGER! |
|
Highly flammable liquid and vapor. Splashes will cause serious eye irritation. May cause drowsiness or dizziness if inhaled. Precautions during use: Keep away from heat, sparks, and open flames. No smoking. Keep container tightly closed. Avoid breathing in vapors. Use in a well-ventilated area. Wear eye protection. First aid: If inhaled: Remove person to fresh air and keep comfortable for breathing. Call a doctor if you feel unwell. Eyes: Rinse cautiously with water for several minutes. Remove contact lenses if it is possible. Continue rinsing. If eye irritation continues, see a doctor. Emergency: In case of fire, use water spray, alcohol resistant foam, dry chemical or carbon dioxide to put it out. Manufactured by: Greedist Chemicals Co. 111 Only Drive, Onlyville, Iowa, 11111 USA. |
| CAS #67-63-0 |
But the truth is that nobody knows how dangerous many chemicals are because not many chemicals have been studied fully for their effects on people. It takes a long time to do scientific research and even more to make laws to protect people. And to make it even more complicated, it is even more unusual for scientists to study how a mix of chemicals affects peoples’ health. And how often do you use only one chemical? That is why it is important for companies to use only chemicals that are already proven to be safe.
Read the label
Every chemical container should have a label on it, written in a language people in your factory can understand. If the containers you are working with do not have labels, ask the supervisor to provide you with this information. You can also ask the shipping department workers if they could share with you the information on the label of the larger container the chemical came from, or you can try to find out more information yourself. In some countries, these labels are required by law to provide information in many languages.
Get the Safety Data Sheet (SDS)
Companies that make chemical products publish an information sheet for each product they make. These used to be called Material Safety Data Sheets (MSDS) but are now called Safety Data Sheets (SDS).

An SDS is often long and difficult to understand. Though each sheet must use the same categories, the content in the SDS for different companies’ sheets may be very different, even for the same chemical. To get more information, read several SDS from different manufacturers of the same chemical.
How to Get and read an SDS
The factory administration should have an SDS for every chemical used in the factory. Your boss should make copies of these SDS available to you and other workers in your language (see The right to know about chemicals).
While you organize to make your boss provide you up-to-date SDS, you can try to get them in other ways:
- Ask the workers who receive, sign for, and store the chemical containers if they have the SDS and could get you a copy.
- Find the name and contact information for the company that produces the chemical from the label on the container and request they send you an SDS in your language.
- Ask staff in unions, worker centers, environmental organizations, or universities for help in finding the SDS or chemical information.
- Research the chemical on the Internet. Search by the name and the CAS number. Look on websites of the companies that produce the chemicals as well as sites that provide SDS from many sources. Compare the different SDS, they might have different information!
In Appendix B you will find links to websites where you can find more information about chemicals. But many of the websites with chemical information are as hard to read as SDS themselves!
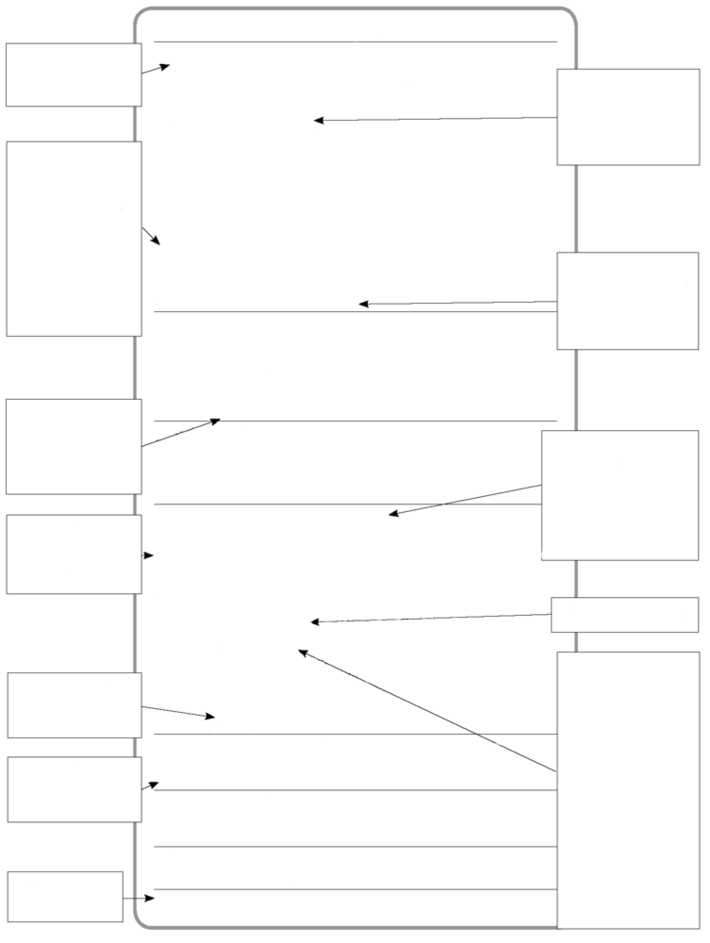
Component information: This product is considered to be hazardous according to CFR 1910.1200.
Acute effects: Irritation of the skin and/or upper respiratory tract,
drowsiness, headache.
Chronic effects: Slightly hazardous in case of skin contact (sensitizer).Carcinogenic effects: A4 (Not classifiable for human or
animal.) by ACGIH, 3 (Not classifiable for human) by IARC.
Inhalation: Mild irritation of eyes, nose, and throat.
Ingestion: Drowsiness, headache.
Dermal Contact: Dry, cracking skin.
Seek medical attention.
Skin: Wash with soap and water. Take off contaminated clothing and
shoes. Obtain medical attention.
Inhalation: Remove victim to fresh air. Give oxygen if breathing is difficult.
Seek medical attention.
Ingestion: Do not vomit. Seek medical attention.
Flash point: 12 °C (53.6 °F)
Auto ignition temperature: 339 °C (750 °F)
Fire Hazard: Highly flammable when there is a spark or heat.
CAUTION: MAY BURN WITH NEAR INVISIBLE FLAME.
Explosion hazards: Explosive when there is a spark or heat.
Fire Fighting Instructions: Water may be ineffective. Do not use a solid
water stream because it may spread the fire. Cool containers exposed
to fire or heat with water.
SMALL FIRE: Use DRY chemical powder.
LARGE FIRE: Use alcohol-resistant foam, carbon dioxide,
water spray, or fog.
Large spill: Keep away from heat and sparks. Use dry earth or sand to absorb it.
Ensure all equipment is electrically grounded.
Storage Recommendations: Keep in a cool area with good ventilation. Keep in
a segregated area. Store in tightly closed containers.
mists. Ground containers to prevent static sparks. Ensure eyewash
stations and safety showers are proximal to work-stations.
and flame retardant antistatic protective clothing.
Eye: Wear safety glasses with side-shields. For leak, spill, or other
emergency, use chemical goggles and face-shield.
Respiratory: NIOSH approved respiratory protection when levels are high.
Full suit. Vapor respirator. Boots. Gloves.
2,000 ppm TWA: 983 STEL: 1230 (mg/m3) [Australia] TWA: 200
STEL: 400 (ppm) from ACGIH (TLV) [United States] [1999]
Odor: Pleasant. Odor resembling that of a mixture of ethanol and acetone.
Taste: Bitter (slightly).
Color. Colorless.
Odor Threshold: 22 ppm (Sittig, 1991) 700 ppm for unadapted panelists
(Versch, 1983)
Conditions to avoids: Heat, ignition sources, incompatible materials
Incompatibilities: Reacts violently with hydrogen + palladium
combination, nitroform, oleum, COCl2, aluminum triisopropoxide, oxidant
LD50 – Route: Oral; Dose: 4396 mg/kg
LD50 – Route: Ingestion; Dose: 12,800 mg/kg
Harmful by inhalation, if swallowed. Causes headaches and other effect to
nervous system.
kidneys and liver.
effects (fertility, fetoxicity, developmental abnormalities based on animal
studies. Detected in maternal milk in human.)
[Fathead Minnow]. 64000 mg/l 96 hours [Fathead Minnow].
Teratogenic: causes birth defects.
Fetoxicity: can damage baby in the womb.
Development toxicity: can affect the baby’s development.
regulations.
Consider fuels blending as an alternative to incineration.
Community resources
Labor unions, women’s groups, and environmental organizations may be able to help you get information. If you know the name of a chemical, you can find information in libraries and on the Internet. But even if you do not know the name, you can sometimes find out the name with other information, such as use, color, smell, and so forth. Any information can be helpful.
We demand to know what chemicals
are being used in the factory
In the early 1980s, a group of workers, environmental activists, and community members in New Jersey, USA got together to demand that the government pass the "Right to Know" law. This law would give workers the right to know what chemicals were used in their workplaces.
Workers and their unions had long demanded that employers tell them exactly what chemicals they worked with. But employers fought back, with the law on their side. Even after workers got rashes or had trouble breathing, employers didn’t have to tell workers what was in the mixes. They said, "If we tell workers or health inspectors, our 'trade secrets' will be known and we won’t be able to compete." They would rather let workers die than disclose those chemicals.
Although workers were at the front line of chemical exposures, they were not the only ones getting sick from chemicals they knew nothing about. Pollution of air and water, burial of toxic waste in the community, and accidental toxic releases and fires were exposing people in New Jersey to all kinds of chemicals. And they were getting angry, too!
Connecting workers inside the factory with people outside was a very successful strategy. It brought together activists from different sectors: mothers, politicians, environmentalists, and union members, all under one single banner: We have the Right to Know!
Environmental crises pushed even more people to support and organize for a new law. In 1983, the Right to Know bill was passed.

The Right to Know law was a big advance, but it has not protected workers and the community enough from chemical exposures. While industry attacks the law, people continue to organize for public disclosure and safe handling of chemicals used at work, for safe disposal of chemicals, and for policies to stop accidents from happening and training to handle them when they do.



